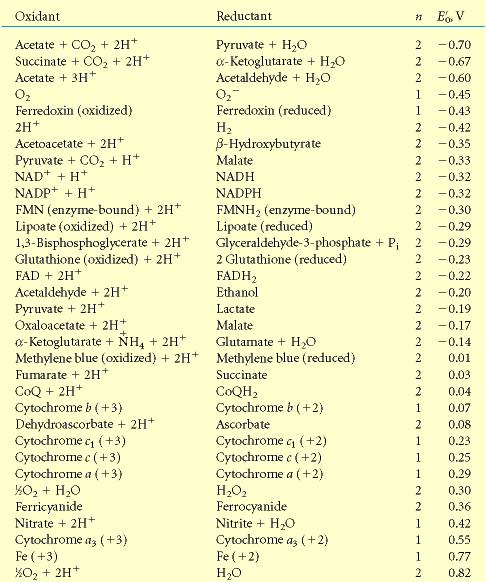
Exam 3
Answer the following. Physical constants and a table of standard reduction potentials are located on the last page of the test. Graph paper is attached. Assume all temperatures are 37o C. There are 103 points on this test. Good Luck.
1)
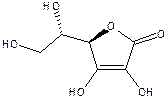 Ascorbic acid (Vitamin C, below) acts
as an antioxidant in vivo, helping protect the body against oxidizing. mutation-inducing chemical agents as well as against
radiation. Oxidized ascorbate
(dehydroascorbate) isn’t much use, however. Plants can use NADPH to reduce the oxidized
form of ascorbate (dehydroascorbate)
back to ascorbic acid as shown in the reaction below:
Ascorbic acid (Vitamin C, below) acts
as an antioxidant in vivo, helping protect the body against oxidizing. mutation-inducing chemical agents as well as against
radiation. Oxidized ascorbate
(dehydroascorbate) isn’t much use, however. Plants can use NADPH to reduce the oxidized
form of ascorbate (dehydroascorbate)
back to ascorbic acid as shown in the reaction below:
Dehydroascorbate + NADPH + H+ ⇌ Ascorbate + NADP+
What is the equilibrium constant for this reaction? (Standard reduction potentials are on the last page) (6 pts)
|
Dehydroascorbate + 2e- + 2 H+ |
⇌ |
Ascorbate |
+0.08 V |
|
NADPH + H+ |
⇌ |
NADP+
+ 2e- + 2 H+ |
+0.32 V |
|
Dehydroascorbate + NADPH + H+ |
⇌ |
Ascorbate
+ NADP+ |
+0.40 V |
![]()
![]()
2) The Krebs cycle produces both NADH and FADH2. Both transfer their electrons to the electron transport chain and becomes reoxidized. Each NADH produced in the Krebs cycle results in the production of (optimally) about 2.5 - 3 ATPs, while each FADH2 produced in glycolysis only results in (optimally) about 1.5 - 2 ATPs. Explain why this is true. (5 pts)
Electrons
from FADH2 enter ETC at Complex II, skipping complex I (NADH
Dehydrogenase) and therefore pump fewer protons across the inner mitochondrial
membrane.
3)
You have the good fortune to be
responsible for examining the biochemistry of an organism recovered from the
subsurface
![]()
![]()
DY = .081 V = 81
mV
4) We mentioned the fact that, under low-oxygen conditions, yeast and some other microorganisms convert the pyruvate produced in glycolysis into ethanol. Similarly, mammals under oxygen stress will convert the pyruvate to lactate. Why do they do that? (5 pts)
\NAD+ is a
required reactant in the glyceraldehyde-3-phosphate dehydrogenase step of
glycolysis. In the absence of oxygen,
NAD+ cannot be regenerated from NADH by electron transport. Instead, NADH is used to reduce pyruvate.
5) Here it comes: diagram the reactions of glycolysis, including structures of the intermediates, enzymes, and products. (24 pts)

6) Differentiate among active transport, facilitated diffusion, and passive (i.e. plain old ordinary) diffusion. How do they differ? Why would I ever want to perform active transport in the first place? And how can I tell the difference between facilitated and passive diffusion? (8 pts)
Facilitated
diffusion and passive diffusion both allow materials to cross the membrane in
response to their concentration gradients.
Passive diffusion involves molecule to which the membrane is permeable;
facilitated diffusion involves the passage of molecules to which the membrane
is impermeable through
channels or transport molecules. As
such, they require no additional input of energy. Active transport pumps materials against
their concentration gradients and requires energy to proceed. Many materials need to be maintained in
disequilibrium across the membrane – e.g. Na+ and K+ ions.
Facilitated
diffusion can be distinguished from passive diffusion in that it is saturable.
7) Match the following compounds with their descriptions (3 pts each):
(a) OH _C__ wax
HC-CH=CH-(CH2)12-CH3
|
O _d__ saturated fatty acid
HC-NH-C-(CH2)8-CH=CH-(CH2)5-CH3
|
_a__ ceramide
CH2OH
_b___ phosphatidylcholine
+ CH3 O
(b) H3C-N-CH2-CH2-O-P-O-CH2 _e___
triacylglycerol
CH3 O
| O
HC-O-C-(CH2)16-CH3
| O
HC-O-C-(CH2)16-CH3
H
O
(c) H3C-(CH2)7-CH=CH-(CH2)7
-C-O-(CH2)8-CH=CH-(CH2)7-CH3
O
(d) HO-C-(CH2)18-CH3
H
O
(e) HC-O-C-(CH2)14-CH3
|
O
HC-O-C-(CH2)14-CH3
|
O
HC-O-C-(CH2)14-CH3
H
8) Define the following terms in one or two sentences MAX!!! Provide enough info so that I know that you know what you are talking about; however, the “Great American Novel” answer will be penalized! (4 pts each)
a) Decoupler (or uncoupler) (Hint: proton gradient)
A decoupler breaks down the proton gradient across the inner mitochondrial membrane. Electron transport proceeds, bu the disrupted proton gradient interferes with ATP synthase activity.
b) Ubiquinone (also known as Coenzyme Q)
Electron carrier in the IMM which transfers electrons from complexes 1 and 2 to complex 3. Can carry 2 protons and 2 electrons.
c) ATP Synthase
Multienzyme complex anchored in the IMM. F0 portion is a membrane-bound rotor driven by the H+ gradient across the IMM. F1 portion is driven by the F0 rotor and synthesizes ATP fro ADP and Pi.
d) G-Protein
Multi-subunit protein associated with cell surface receptors. Binding of a hormone by the receptor triggers alpha subunit of the G protein to exchange ADP for ATP. ATP-bound form dissociates from the beta/gamma chains and activates membrane-bound enzymes like Adenylyl cyclase. The alpha subunit is also a slow ATPase; upon ATP hydrolysis, the alpha subunit returns to its original conformation and reassociates with the beta and gamma subunits.
e) Glycogen Phosphorylase
Enzyme responsible for releasing glucose-1-phosphate from the non-reducing ends of glycogen.
z
f) Futile cycle (hint – exploding patients)
Loss of reciprocal control between PFK-1 and fructose-1,6-bisphosphatase, resulting in the generation of massive amounts of heat with neither net oxidation of glucose nor net synthesis. One cause is exposure of susceptible individuals to halothane anaesthetics.
g) Cori cycle
Glucose in the muscles is converted to pyruvate by glycolysis. Lactate created from pyruvate in the muscles in order to regenerate NAD+ is transported in the bloodstream to the liver, where it is reoxidized to pyruvate. Pyruvate enters the gluconeogenic pathway, where it is used to make glucose. The glucose leaves the liver in the bloodstream and is delivered to the muscles.
Physical Constants and Parameters
|
Constant |
Value |
|
Avagadro’s Number |
6.02 * 1023 |
|
Boltzmann’s Constant |
1.3807 * 10-23 JK-1 |
|
Charge on electron |
-1.602 x 10-19 coulomb |
|
Gas Constant (R) |
8.314 JK-1mol-1 |
|
Faraday’s Constant |
96,485 J/Vmol |
|
k = 1/(4pe) |
8.99 Nm2coulomb-2 |
|
ln x |
2.303 log x |
Standard Reduction Potentials: