CH 339K Problem Set 1
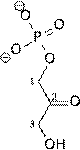
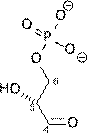
1) Dihydroxyacetone
phosphate (DHAP)and Glyceraldehyde-3-Phosphate (G3P) are both
intermediates in the metabolic breakdown of glucose. They can be interconverted by the enzyme Triose Phosphate Isomerase.
|
|
|
|
|
DHAP |
|
G3P |
For the
conversion of DHAP to G3P, DGo
= 7.5 kJ/mol
a) Calculate
Keq and the equilibrium fraction of G3P at 37o C.

b) What is DG if [G3P] = 1/100 [DHAP]?
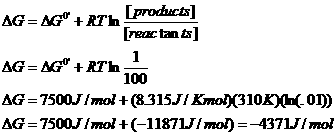
2) A
Cl- ion is separated from a Na+ ion by a distance of 0.5
nm. What is the interaction energy (in
Joules/mole) in
a. Water
(ε = 78.54)
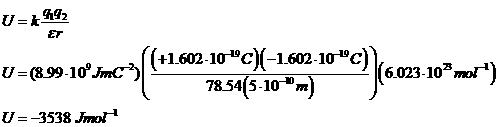
b. n-Pentane
(ε = 1.84)
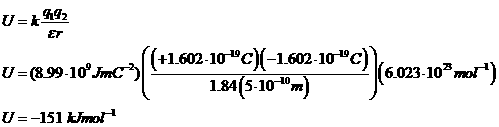
The Point: electrostatic interactions are stronger in environments with lower dielectric constants (like, say, organic solvents or the hydrophobic interiors of macromolecules.)
3) What
is the pH of each of the following unbuffered solutions?
a. 0.35
M HCl
HCl is a strong acid, so it
dissociates completely to (formally speaking) H+ and Cl-.
pH = -log[H+] = -log(0.35) = 0.456
b. 0.35
M Acetic Acid
HAc is a weak acid: we can solve using the
quadratic equation:
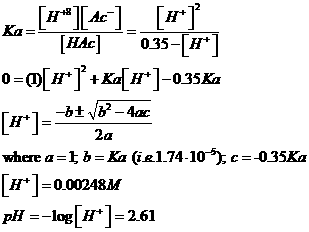
or the approximation:
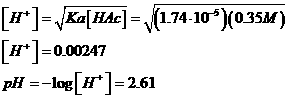
c. 0.035
M Acetic Acid
To heck with the quadratic equation:
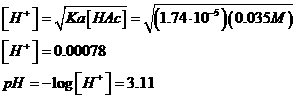
4) What
is the pH of the following buffer mixtures?
d. 1
M Acetic Acid and 0.5 M Sodium Acetate?
Conjugate acid is HAc; conjugate base is Ac-; Na+ is spectator.
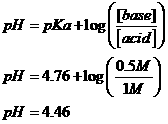
e. 0.3
M H3PO4 and 0.8 M KH2PO4?
Conjugate acid is H3PO4; conjugate base is H2PO4-; K+ is spectator.
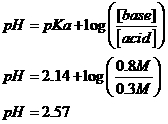
5) You
want a buffer of pH 7, made using KH2PO4 and Na2HPO4:
f. What
is the conjugate acid in this system?
What is theconjugate base?
Conjugate acid is H2PO4-;
conjugate base is HPO4-2
g. If
the buffer is 0.1 M KH2PO4, what must the concentration
of Na2HPO4 be?
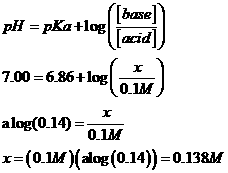
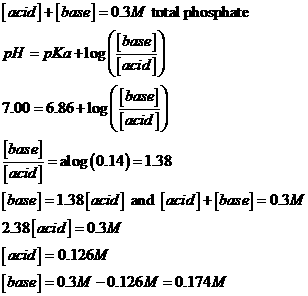
h. You
want the same buffer, but with a total phosphate concentration of 0.3 M. What concentrations of each species do you
need?
6) You
have a beaker containing 500 ml of 0.10 M potassium formate buffer pH
3.75. What is the pH if you add 5 ml of
1.0 M KOH? (Hint – the change in volume
can be ignored)
The conjugate acid is formic (HCOOH, pKa = 3.75); the conjugate base is formate ion (HCOO-). Adding the strong base, KOH, will convert formic acid to formate, changing the base/acid ratio and changing the pH.
First, how much of each species do we have in our starting solution?

Now, when we add 5 ml of 1M KOH, we
are adding ![]() strong base. This will
convert 0.01M HCOOH to 0.01M HCOO-.
The new pH then becomes:
strong base. This will
convert 0.01M HCOOH to 0.01M HCOO-.
The new pH then becomes:
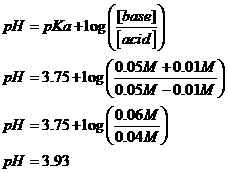
7) On
my benchtop, I have a jar of the amino acid glycine, a solid powder with MW =
75.07 g/mol. I also have a bottle of 1 M
NaOH, all the distilled, deionized water I can use, and a 2 liter graduated
cylinder. Tell me how to make 2.0 liters
of 0.1 M glycine buffer, pH 9.
The conjugate acid is glycine (H3N+CH2COO-), the conjugate base is gly- (H2NCH2COO-).
How much glycine do I need?
![]()
Now, how much strong base do I need to add to convert the correct amount of glycine to gly- to give the correct conjugate base/conjugate acid ratio to generate a pH of 9:

In order to convert 0.02M of the free acid (glycine) to the conjugate base (gly-), I have to add 0.02M strong base (KOH)
![]()
So I need to mix 15.14g glycine, 40 ml of 1M KOH, and add water to 2 liters total volume.
Useful Physical Constants
|
Constant |
Value |
|
Avagadro’s Number |
6.02 * 1023 |
|
Boltzmann’s Constant |
1.3807 * 10-23 JK-1 |
|
Charge on electron |
-1.602 x 10-19 coulomb |
|
Gas Constant (R) |
8.314 JK-1mol-1 |
|
Faraday’s Constant |
96,485 JV-1mol-1 |
|
k = 1/(4pe) |
8.99 x 109 Nm2coulomb-2 |
|
ln(x) |
2.303 log (x) |
|
Dielectric constant of water |
78.54 |