Oxygen Binding Proteins I: Myoglobin
Okay, we’ve looked at the structural features of proteins; now let’s examine the properties of one group of proteins that will introduce us to some general features of how proteins behave and function.
|
Helix |
Residues |
|
A: |
S3 - E18 |
|
B: |
D20 - S35 |
|
C: |
H36 - K42 |
|
D: |
T51 - A57 |
|
E: |
S58 - K77 |
|
F: |
L86 - T95 |
|
G: |
P100 - R118 |
|
H: |
G124 - L149 |
If you are a mammal, every once in a while you need to run away from something really scary – a lion, a bear or a big ugly guy with a gun. At such times you may be using oxygen in your muscles faster than you can breathe. Organisms have several ways in which to deal with this potentially serious problem. One way involves the presence in muscles of an oxygen storage protein called myoglobin. (“Myo” refers to muscle, “globin’ refers to a globular protein.)
Sperm whale myoglobin was the first protein to have it’s three-dimensional structure determined; the muscle tissue of deep-diving whales is a rich source of myoglobin. Sperm whale myoglobin is a protein comprised of 153 amino acids with a molecular weight of about 14,000. There are 8 a-helices, designated A – H; the remaining non-helical parts of the molecule are bends and turns between the helices. The helical portions are shown in the table.
 In
addition to the amino acids, the molecule also contains a prosthetic
group (an non-protein component attached to
the protein structure that assists it in functioning) called heme. Heme consists of a large polycyclic
organonitrogen compound derived from porphyrin, plus a ferrous ion, Fe+2,
covalently coordinated to four nitrogen atoms in the porphyrin system. The ferrous ion is also coordinated to the
nitrogen of a histidine side chain in the protein, His 63 (?). The combination of the iron and the highly
conjugated porphyrin ring makes heme red; myoglobin is the primary reason for
the redness of red meat.
In
addition to the amino acids, the molecule also contains a prosthetic
group (an non-protein component attached to
the protein structure that assists it in functioning) called heme. Heme consists of a large polycyclic
organonitrogen compound derived from porphyrin, plus a ferrous ion, Fe+2,
covalently coordinated to four nitrogen atoms in the porphyrin system. The ferrous ion is also coordinated to the
nitrogen of a histidine side chain in the protein, His 63 (?). The combination of the iron and the highly
conjugated porphyrin ring makes heme red; myoglobin is the primary reason for
the redness of red meat.
Another histidine, His 94 (?) lies above the plane of the heme but is too far away to form a bond to the iron. Instead it forms the roof of a pocket that lies above the heme and into which dissolved O2 molecules can diffuse. O2 can then reversibly form a sixth bond with the heme iron. Interestingly, if free heme is exposed to O2, the iron atom is readily oxidized from ferrous (Fe+2) to ferric (Fe+3). Bound into the hydrophobic environment of the protein interior, however, the iron can bond to oxygen without itself becoming oxidized.
A molecule similar in shape and structure to O2 is CO – carbon monoxide. CO binds readily to the heme iron, but the binding is essentially irreversible. Once CO is bound, the myoglobin molecule (and it’s sister hemoglobin molecules in the blood) are no longer capable of binding and releasing oxygen; this is the basis of carbon monoxide poisoning, a common form of suicide as well as accidental death. When heme binds oxygen it changes color and becomes redder than heme without bound oxygen. Heme with CO attached is also a brighter red than unbound heme. This accounts for the well known phenomenon whereby people who have died of carbon monoxide poisoning generally have bright red cheeks and look, if anything flushed and healthy – especially when compared to other corpses.
Okay, let’s take a closer, more quantitative look at O2 binding. We can describe the reaction between myoglobin, which we will abbreviate Mb, and O2 as follows;
Mb + O2 ⇌ Mb· O2 (1)
Using our knowledge of general chem., we can define an equilibrium constant for the reaction such that:
![]() (2)
(2)
Solving for [Mb· O2] we get:
![]() (3)
(3)
Okay, let’s assume we measure O2 binding by myoglobin in terms of fractional binding – that is, what fraction of the available myoglobin is in the bound state. We can define a term Q such that:
![]() (4)
(4)
If we substitute (3) for [Mb·O2], then we get:
![]() (5)
(5)
which simplifies to;
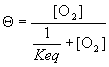 (6)
(6)
Now, there exists some oxygen concentration, some [O2], where Q is 0.5 – that is, half the myoglobin exists in the unbound state. If we designate this value of [O2] as [O2]50 then we can write equation (6) as:
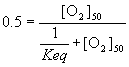 (7)
(7)
And for this to be true, ![]() must be equal to [O2]50 . So we
can now rewrite (6) as:
must be equal to [O2]50 . So we
can now rewrite (6) as:
![]() (8)
(8)
Thinking back to General Chemistry one more time, we recall that the concentration of gas in a solution is equal to the partial pressure of that gas over the solution. We can therefore substitute the partial pressure of oxygen, pO2, for the concentration, [O2]. We can also substitute the partial pressure of oxygen where Q = 0.5, p50, for [O2]50. Equation (8) then becomes;
![]() (9)
(9)
This generates a hyperbolic binding curve. For sperm whale myoglobin, p50 is approximately 4 mm Hg. Using Equation (9), we generate the values and curve below, which closely approximate experimental data.
|
pO2
|
q |
|
0 |
0.000 |
|
3 |
0.429 |
|
6 |
0.600 |
|
10 |
0.714 |
|
30 |
0.882 |
|
60 |
0.938 |
|
100 |
0.962 |

Myoglobin thus makes an excellent oxygen storage protein, The partial pressure of oxygen in the lungs is generally in the range of 100 mm Hg; the partial pressure in the peripheral bloodstream is generally around 30 mm Hg. Therefore myoglobin is always capable of extracting oxygen form the blood and storing it in muscle tissue pending need. When the dissolved oxygen concentration of the muscle tissue drops low during exertion, the oxygen can be released to feed energy production.