CH339K
Chemical
Bonding
In
order to maintain their existence and carry out the complicated sets of
reactions they require, biological systems need to
establish and maintain molecular integrity, to maintain higher-order
structures, and to bring together multimolecular assemblages of varying
specificity and duration. In order to
meet these different requirements, organisms use every available type of
interatomic interaction: covalent bonding, and the full range of noncovalent
charge interactions. We will concentrate
on five “bond” types:
·
Covalent bonds
·
Ionic bonds
·
Dipole interactions
·
Hydrogen bonds
·
Hydrophobic effects (we’ll discuss in conjunction with water chemistry)
Covalent Bonding
The
nature of the covalent bond is covered in General Chemistry; we will touch on
it only briefly. Covalent bonds result
from the rearranged orbitals which the bonding electrons assume around the
nuclei of the bonded atoms. The strength
of the interaction results from the attraction of the positively charged nuclei
to this joined electron “cloud” and is normally quite high. Covalent bond energies are normally in the
range of 25 – 110 kcal/mol
( 100 – 475 kJ/mol ).
|
Atom |
Bond
Number* |
|
C |
4 |
|
H |
1 |
|
O |
2 |
|
N |
3 |
|
P |
3,5 |
|
S |
2 |
*the
bond numbers illustrated are typical of biological systems and should not be
considered set in stone!
The
strength of the covalent bond depends on a number of factors; for example, as
previously discussed, repulsion between lone electron pairs on adjacent
nitrogen atoms results in a relatively weak ( 25
kcal/mol ) N-N bond. As we’ll discuss
soon, the common currency for energy transfer within a cell is the cleavage of
a triphosphate to orthophosphate and pyrophosphate. The electrostatic repulsion that exists
between negatively charged oxygen atoms in the phosphate chain destabilizes the
bonds between them and makes them readily subject to hydrolysis. Commonly referred to in textbooks as “high –
energy” phosphate bonds, these bonds are actually rather low – energy structures. If
they were really high – energy bonds, they would be difficult to break and
would not be key participants in so many reactions! The “high – energy” misnomer comes from the
relatively large amount of free energy recovered from their hydrolysis.
Ionic Bonds:
Ionic
bonds, resulting from the attractive force between oppositely charged atomic
and / or molecular ions, are just one ( strong )
extreme of a series of electrostatic interactions involving complete or partial
charges. The force of an ionic interaction follows Coulomb’s Law:
![]()
The
constant k is
related to the permittivity of the vacuum, and is equal to:
![]()
q1, q2 are the
respective charges in coulombs (for 1 electron, q = 1..6
·10-19 coulombs ).
r
is the distance separating the charges in meters.
e is
the dielectric constant of the medium, in dimensionless units.
The
dielectric constant for a medium is a measure of it’s
own proclivity to engage in charge interactions and thus represents a shielding
effect on the charges by the medium itself.
The dielectric constant of the vacuum is defined as 1. The dielectric constant of water is very
high, about 80. Most organic solvents
have dielectric constants much less than water.
The interior of a protein is a very poor dielectric, about 4, and charge
interactions in such a locale are quite strong.
We
are usually more directly concerned with the energy of the interaction than
with the force. In the case of ionic
interactions, the “bond” energy is the energy required to completely bring the
two charges together from infinite distance to their current position. We thus get:
![]()
It
can thus be seen that ionic interactions are fairly long-range effects, falling
off as a function of the distance between the charges. Ionic interactions are also
non-directional. Charge – charge
interactions within and between macromolecules are often referred to as salt links, since the association of a
positive charge, say an amino group, with a negative charge, say a carboxyl
group, mirrors the association of anions and cations into a salt.
The
strength of ionic interactions is quite dependent on the medium. For example, in a vacuum, the strength of the
“bond” between, say, a positive amino group and a negative carboxyl group
separated by 4 Å is about 80 kcal/mol, equivalent to a fairly
strong covalent bond. This
explains the high melting and boiling points of ionic solids. In the interior of a protein, however, from
which water is excluded, the strength of the same interaction is about 20 kcal/mol. On
the fully exposed and solvated surface of a molecule, the same interaction has
a strength of only about 1 kcal/mol.
Dipole Interactions (van der
Waal Interactions) :
When
electrons are shared unevenly between covalently bonded atoms, the result is a
dipole – each atom has a partial negative or positive charge. Unequal electron distribution results from
the different relative abilities of different atom types to attract
electrons. This power to attract
electrons is measured by the electronegativity
scale.
|
H = 2.1 |
|
|
|
|
|
|
|
Li = 1.0 |
Be = 1.5 |
B = 2.0 |
C = 2.5 |
N = 3.0 |
O = 3.5 |
F = 4.0 |
|
Na = 0.9 |
Mg = 1.2 |
Al = 1.5 |
Si = 1.8 |
P = 2.1 |
S = 2.5 |
Cl = 3.0 |
|
K = 0.8 |
Ca = 1.0 |
Ga = 1.6 |
Ge = 1.8 |
As = 2.0 |
Se = 2.4 |
Br = 2.8 |
|
Rb = 0.8 |
Sr = 1.0 |
In = 1.7 |
Sn = 1.8 |
Sb = 1.9 |
Te = 2.1 |
I = 2.5 |
|
Cs = 0.7 |
Ba = 0.9 |
Tl = 1.8 |
Pb = 1.9 |
Bi = 1.9 |
Po = 2.0 |
At = 2.2 |
The
more electronegative an element, the greater it’s
power to attract electrons. Electronegativities increase as one goes up the periodic
table and to the right. Fluorine is the
most electronegative element (4.0), cesium the least (0.7) The greater the difference in electronegativities in two bonded atoms, the greater the
inequality of electron distribution between them. For example, the electronegativity of oxygen
is 3.45; that of carbon is 2.55, indicating that
oxygen has a higher affinity for electrons than carbon. The carbon – oxygen bond will therefore form
a dipole, with a partial negative charge on O and a partial positive charge on
C. The magnitude of the dipole depends
on
(a) the magnitude of the charge difference (q), and
(b) the separation distance of the partial charges.
That
is: m = q ´ x
In
addition to permanent dipoles, as
mentioned above, the presence of a complete or partial charge in close
proximity to an otherwise non-polar molecule may alter the electron
distribution within the second molecule, producing an induced dipole. For example,
the amino acid glycine normally exists in solution with a negatively charged
deprotonated carboxyl group at one end and a positively charged protonated
amino group at the other. If a glycine
sits above the plane of a benzene molecule, the p - electron cloud of the
benzene ring will be distorted, with the electrons being preferentially drawn
to the end of the ring adjacent to the glycine amino group, and preferentially
repelled withdrawn from the end adjacent to the glycine carboxyl group. The resulting induced dipole will interact
with the glycine zwitterion. Induced
dipole effects are always attractive.
A
special case produces the London dispersion force (frequently referred to by
biochemists as “van der Waal forces”, though this term applies to all
noncovalent electrostatic interactions.)
Any atom will be found at some given instant of time to be a transient dipole,
resulting from the uneven distribution of negative electrons around the
positive nucleus at any given moment.
When atoms are in close proximity, there is a tendency for their
transient dipoles to flicker in unison.
This produces a weak attractive force between them. These dispersion forces are individually
weak, on the order of 0.1 – 1 kcal/mol.
They are, however, the forces responsible for the liquefaction of noble
gases at low temperatures. Also, in
biological macromolecules, the energetic effects of large
numbers of such contacts in molecules with complimentary surfaces becomes
quite large.
As
you might expect, dipoles interact in a manner analogous to ions; the
interactions differ both in their strengths ( partial
charge interactions are weaker ) and the range over which they operate. The energy of charge – dipole interactions
falls of as 1/r2; the weakest interactions, those between induced
dipoles, fall of as 1/r6.
The
effective surface of a molecule depends on the relation between the attractive
dispersion forces and the repulsive effects of electron cloud overlap which
come into play at extremely short distances.
As the distance between charges shrinks, the attractive force becomes
stronger. At very short distances,
however, repulsive forces come into play and rapidly make closer approach
between the two atoms energetically prohibitive. Since the repulsive term varies as 1/r12,
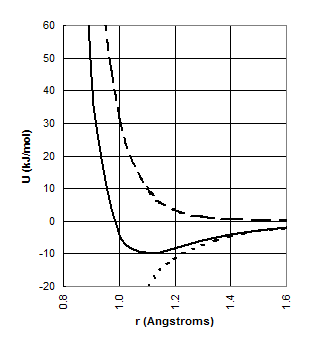
this “wall of energy” goes very large over very short distances and represents
the effective surface of the atom. The
interaction energy at a given radius r is given by the Lennard-Jones
equation:
![]()
where Uo
is the energy minimum – i.e. the strongest possible energy for the interaction
- which occurs at the optimum radius ro. The distance at which energy is minimized is considered the effective
radius and is known as the van der Waals radius of the atom. Van der Waals radii
are on the order of 1 to 2 Å
Hydrogen Bonds:
A
special type of linkage occurs when a hydrogen atom, covalently bound to an electronegative atom, makes a
partial bond with an electron pair on another nearby electronegative atom. Such a bond is referred to as a hydrogen
bond; hydrogen bonds are very important in aqueous,
and especially biological systems.
The
group to which the participant hydrogen is covalently bound is the hydrogen bond donor; the group to which
the hydrogen bond forms is the hydrogen
bond acceptor.
|
Donors |
Acceptors |
|
NH2 |
:N
- |
|
OH |
:O
= |
|
SH |
:O
- |
Some
important points regarding hydrogen bonds
·
Hydrogen bonds are partially
covalent in character. The interaction is more than
electrostatic, as reflected by the interatomic distance between donor and
acceptor atoms, which is considerably less than would be expected from the van
der Waals radii, indicating the actual sharing of electrons.
·
Hydrogen bonds are highly
directional. Unlike purely electrostatic interactions, the
orientation of the participants is critical to bond strength. H – bonds are
strongest when the donor, the shared hydrogen and the acceptor are
collinear. Bond strengths fall off
quickly as the bond is distorted.
·
Hydrogen bond strengths are typically in the range
of 2 – 5 kcal/mol. They are thus of
intermediate strength between covalent bonds and van der Waals contacts.
Water – Properties of
Biological Import:
Above
all, biochemistry is aqueous chemistry.
Most biochemical reactions take place in more – or – less dilute aqueous
solution. As a result, the properties of
water are critical for biochemistry.
Water
is a tetrahedral molecule, with the oxygen atom central and either a hydrogen
or a lone electron pair at each apex of the tetrahedron. Oxygen is highly electronegative. As a result, electron density is enhances
around the oxygen and attenuated around the hydrogens. Water is highly polar
and, as might be expected, is an excellent hydrogen bond donor and hydrogen
bond acceptor. In the liquid state, each
water molecule forms a network of hydrogen bonds with surrounding
molecules. These bonds are constantly
breaking and reforming, with half – times on the order of 10-10
seconds. Time – averaged water molecules
are involved in about 3.5 hydrogen bonds.
This
hydrogen bond network is responsible for many of the biochemically and
biologically critical characteristics of water.
·
Water has abnormally high
melting and boiling points. Methane, ammonia and even the
heavier but structurally similar hydrogen sulfide are all gases at normal earth
surface temperatures. H2S is very
similar to water, but the H-S bond length is greater and O has been replaced by
the much less electronegative S (2.5 vs. 3.5).
As a result, the molecule is a far weaker dipole, does not form hydrogen
bonds, and despite it’s greater molecular weight is a
gas at room temperature.
·
Water has a very high
specific heat
( 1 cal/g. ) It
takes a lot of effort to change the temperature of water. This buffers organisms against environmental changes,
and on a biosphere level helps moderate the planetary climate. The oceans provide an enormous heat
reservoir, and prevent the huge temperature fluctuations that occur on
waterless planets.
·
Water has a very high heat
of vaporization ( 540 cal/g, compared to 263 cal/g for MeOH
and 59 cal/g for CHCl3. )
This permits the use of evaporative cooling and, once again, moderates
planetary climate.
·
Water is an excellent
solvent for ionic and polar substances. The high
dielectric constant ( 80 ) does an extraordinarily
good job of shielding charges from each other.
Compare water to another hydrogen bonder, methane ( e
= 33 ), or with ammonia (e = 15.5.) As a result, the
strength of interaction between ions in an ionic solid drops to very low
values.
·
Water has a high surface
tension. This is of minor biochemical
importance, but biological and physiological effects are significant. A number of organisms live at the air – water
interface on the surface.
In
the frozen state, each molecule forms four hydrogen bonds ( 2
donated, 2 accepted .) Molecular
rotations permit a fair degree of freedom in the spatial arrangement of the
molecules, and crystalline ice is rather disordered with a fair amount of empty
space in the crystal. This results in
another often overlooked, but critical property of water.
·
The density of water
decreases on freezing. The increase in hydrogen
bonding increases the space between individual molecules (
locking the O – H – O distance at 2.76 Å. ) In addition, the loose crystal structure
contains empty space. As a result, there
are fewer molecules per unit volume in normal ice than in liquid water. The upshot of all this is that ice floats.
Were
ice more dense than water, bodies of water would
freeze from the bottom up, destroying aquatic life. Instead, water bodies freeze on the surface,
leaving an insulating layer between the cold atmosphere and liquid water
beneath.
Hydrophobic Interactions:
A
corollary of the solvent properties of water for polar compounds is that water
s a poor solvent for nonpolar compounds.
This hydrophobic effect is entropically driven.
Water
in the liquid state has a high degree of conformational freedom. When an attempt is made to dissolve a
nonpolar compound, the hydrogen bond network is disrupted. In order to solvate the molecule, water
molecules form a ( relatively )highly – ordered cage In bulk water, the intermolecular forces on
an individual water molecule are isotropic – they come equally from all sides;
at the interface between a nonpolar molecule and water, the forces are
anisotropic. The water molecules are
constrained both translationally and rotationally. These constraints can be minimized by
minimizing the contact between water and the nonpolar substance. Minimization occurs when the nonpolar
material assumes a shape with the minimum surface area. Depending on the conditions, this will be a
sphere or a separate layer. Biological
molecules generally fold in such a way as to minimize contact between nonpolar groups and the aqueous solvent. The interiors of proteins are generally
characterized by close contacts among hydrophobic groups; while the nonpolar interactions among aliphatic
chains of fatty acids forms the basic structure of all biological
membranes.